Evan Luxon has been steadily focused on building medical device companies from the outset of his education and career.
Luxon is an Omaha native, and earned a degree in mechanical engineering at UNL. He has always had an entrepreneurial itch and throughout his education he was drawn to the vast opportunity for innovation in medical devices to have a positive impact on people’s lives. He decided to focus his career on medical device development and framed the rest of his academic experience around that goal.
Luxon attended Johns Hopkins for his master’s degree in engineering with an emphasis in bioengineering innovation and design. He immersed himself in hands-on training by observing surgeons in the OR, identifying unmet clinical needs, and developing a patented product and business plan with a team of other students.
The next stop for Luxon was Stanford for his master’s degree in mechanical engineering.
“Stanford is the academic epicenter for early stage medical device development, and so I deliberately soaked up all I could both through coursework and by consulting for startups on the side, then joined TheraNova, a medical device incubator in San Francisco,” said Luxon.
Luxon is now one of five partners at TheraNova and has been involved in several of the companies it has started, helping nurture the technologies to market for the benefit of patients. Luxon joined one of those companies, Potrero Medical, as its technical co-founder. He led the development of a device at Potrero to improve urine output measurements in the ICU.
“The urine collection units in use were very outdated, required manual charting, and were not effective in draining all the urine from the patient, which is critical for understanding their kidney health. A core piece of the Potrero technology ensures that all urine a patient is producing makes it into the collection bag. Our device brings digital intelligence to this aspect of patient care, aiding providers with greater accuracy and efficiency in tracking their patients’ urine output.”

The company’s device quickly caught the attention of cardiothoracic surgeons using it in the OR. The surgeons asked whether a similar solution could be developed for chest drains. The surgeons cited clogged tubes as a significant problem for patients in recovery following cardiac surgery.
Luxon talked to more surgeons and dug into the medical literature; he found that the problem was pervasive and not only anecdotal. The Potrero board decided the opportunity was ripe to spin out a new company to build a drainage solution for post-operative cardiothoracic care, with Luxon leading the company as co-founder and CEO.
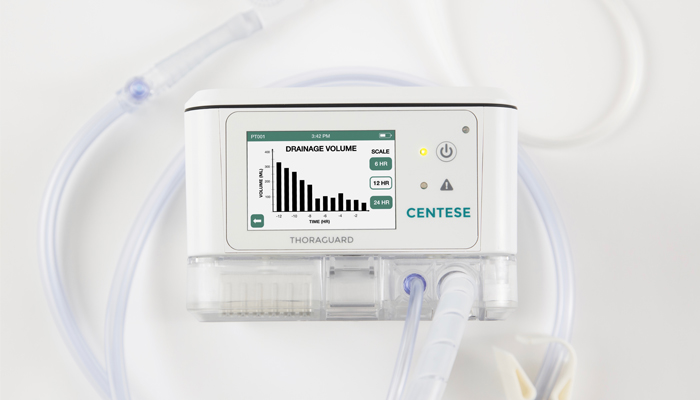
Luxon wrapped up product development at Potrero and made the move back to Omaha in 2015. His company secured grants from the National Science Foundation and the State of Nebraska totaling $1.3 million early on. The company leveraged the financing to raise an additional $2.9 million Series A round in 2017 to complete development of its product, Thoraguard, which automates post-surgical chest tube management in order to improve patient outcomes and reduce costs. Upon receiving FDA clearance for commercial use in 2018, the company has moved forward with pilot studies at premier institutions around the country and is turning its attention to its scaling.
Centese employs seven full time. All are in Omaha with the exception of one at TheraNova, and they span the areas of engineering, quality, and manufacturing. Centese’s device is built on site, including sterile disposable units that are assembled in the company’s ISO Class 7 cleanroom.
“I’ve kept close ties with the team at TheraNova and continue to leverage the expertise there that’s been so valuable to Centese and has contributed to so much of our success in Nebraska.”
Luxon added that his experience founding his company in Nebraska has been very positive.
“Since moving back, people in the startup ecosystem here have been incredibly supportive, which led me to Pipeline, and a strong relationship with UNeMed; a small group of us are networking to establish a medical device startup community in Nebraska that can foster the growth of more medical device companies here.”
UNeMed is the technology transfer and commercialization office for the University of Nebraska Medical Center and the University of Nebraska at Omaha.

Luxon commented on his decision to build a medical device company in Nebraska and the opportunity for a medtech ecosystem to develop here.
“Although certain aspects of medical device development require highly-specialized skills, I believe much of it can be learned through on-the-job coaching. For example, I’ve recruited UNL engineering graduates with exceptional technical skills and helped them learn the intricacies of medical device development, such as how to approach device verification and validation. That said, it’s important for new entrepreneurs in this industry to seek mentors and domain experts to address the challenges specific to medical devices. Many in this space are practicing physicians who see problems in practice and create solutions to solve them, which is an excellent founding perspective when paired with the other necessary expertise, including around regulations and reimbursement.”
Luxon added that aspiring medical technology companies should not necessarily be deterred by regulatory challenges.
“Medical device regulation is commonly misunderstood as being an immense hurdle that takes years and years; however, different devices receive varying levels of regulatory scrutiny. At Centese, for example, we obtained FDA clearance in five months. My main recommendation is to seek someone with FDA expertise as early as possible in order to effectively tailor your product development to meet the regulatory requirements. At Centese, we received this help through our relationship with TheraNova, which has a former FDA Branch Chief on staff. Second, engage with the FDA early as well. There are specific mechanisms in place to have conversations prior to filing – this is tremendously helpful.”
Luxon noted that many opportunities exist in medical devices right now – especially in digitization of medicine, though he pointed out that digitizing does not necessarily make the device more valuable to patients.
However, with respect to Centese, Luxon said, “Digitization in this case creates added value because the current approach to surgical drainage leads to unnecessary complications and costs due to problems that can be solved with better technology. It’s a prime opportunity for us to rethink what’s possible. We believe the drainage market has been overlooked and undervalued historically, and we’re excited to turn that around with far better technology for the benefit of patients and providers.”
Visit http://www.centese.com/ to learn more about this company.